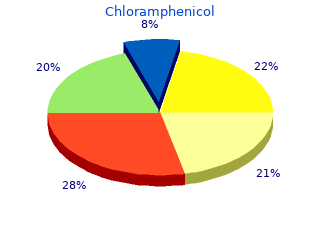
Chloramphenicol
2019, Eastern Michigan University, Bogir's review: "Chloramphenicol 500 mg, 250 mg. Best Chloramphenicol no RX.".
Examples of drug efects augmented in this manner are buy chloramphenicol 250mg on line, postural hypotension with agents that lower blood pressure chloramphenicol 250 mg without prescription, dehydraton, hypovolemia, and electrolyte disturbances in response to diuretcs, bleeding complicatons with oral antcoagulants, hypoglycemia with antdiabetcs, and gastrointestnal irritaton with non-steroidal ant-infammatory drugs. Psychotropic drugs but also antconvulsants and centrally actng anthypertensives may impede intellectual functons and motor coordinaton. The antmuscarinic efects of some antdepressants and neuroleptc drugs may be responsible for agitaton, confusion, and delirium in elderly. If drug therapy is absolutely necessary, the dosage should be ttrated to a clearly defned clinical or biochemical therapeutc goal startng from a low inital dose. Storage The term used to describe the safe keeping of all fnished drugs and pharmaceutcals awaitng dispatch. The term is also applied for safe stores in hospitals and dispensaries under the specifed conditons, so as to maintain their quality and potency. Dosage Form Refers to the gross physical form in which a drug is adminis- tered to or used by a patent. Drug Product A dosage form containing one or more actve therapeutc ingredients along with other substance included during manu- facturing process. Finished Product A medicinal product which has completed all stages of manu- facture including packaging. Strength The concentraton of the drug substance (for example weight/ weight, weight/volume or unit dose/volume basis) and the potency i. Stability Degree of resistance to chemical and physical changes, the efcacy of the preparaton must remain constant or change only within the limit specifed by ofcial compendia. Expiraton Date The date placed on the immediate container label of a drug product that designates the date through which the product is expected to remain within specifcatons. Storage Procedure and Instructons Drugs must be stored under conditons which minimize dete- rioraton, contaminaton or damage. They must be stored under conditons compatble with their recommended storage requirements of temperature and humidity and where neces- sary to comply with legal requirements, under secured or segregated conditons. Appropriate storage conditons are: Temperature or humidity controlled environment must be equipped with suitable indicators, recorders and/or failure warning devices which must be checked at appropriate inter- vals and the results are coded. Temperature should be measured at diferent levels in the warehouse and if necessary storage of sensitve drugs should be restricted to locatons in the warehouse where they will be protected from extreme conditons. Temperatures of the refrigerators, deep freezers, and Relatve Humidity in humidity control area as well as general areas of storage at room temperature should be recorded on a daily basis. Storage conditons not related to temperature are indicated in following terms: Drug storage should be regularly checked for cleanliness and good order and for misplaced/deteriorated/out dated stock. Drugs with expired shelf life should be destroyed unless an extension of shelf life is granted following the sats- factory results or re-analysis. Some categories of supplies require special storage condi- tons which include vaccines, narcotcs, and combustbles e. Narcotcs and other controlled substances should be kept in secure locking rooms with only one entrance. Inspecton for Deterioraton Pharmacists should be aware that deterioraton of drug product may happen even before their expiraton. Hence inspecton should include frequent product examinaton to detect signs of product deterioraton which difer according to dosage form. The Pharmacists in the Stores should prepare an exhaustve list of following deterioraton/spoilage indicators and keep them. Liquid Dosage Forms Slight gradual discolouraton, Swirly precipitaton, Whickering: pin hole at ampoule tp that leaks soluton which precipitate or crystalline solid mater, clouding, fading of colour, Cake sedimentaton (suspension), Creaming and cracking (emul- sion), Discolouraton. Semisolid Dosage Forms Ointments creams, gels and suppositories -Change in consist- ency and feel to touch, Phase separaton, Discolouraton, Surface crystal growth 3. Solid Dosage Forms Surface chipping or pitng (plain tablets), Deformaton (capsules), Increased hardness, Discolouraton, Colour fading (coloured tablets), Chipping of coat (coated tablets). Most vitamins, hormones enzymes are highly sensitve to oxidaton and photo decompositon. The integrity of packaging of dosage form is one of the impor- tant tasks of inspecton for pharmacist as these protect the drug in a tailored fashion. Afer each inspecton, products showing any signs of instability should be subjected to sample analysis to ensure quality. Aerosols Aerosols should be stored in a clean separate area away from heat and sunlight because the container contents are under pressure, flled containers must be checked for weight loss over the expiraton datng period, for contents under pres- sure. The label should display “Do not expose to heat or store at a temperature above 40⁰C, keep out of reach of children”. Creams Creams can be destroyed under extreme temperature fuctua- tons hence they should be stored at temperature above 10oC and not exceeding 30⁰C. If the creams are opened and diluted they should not be kept for more than 14 days to avoid micro- bial contaminaton. Ophthalmic Solutons and Drops They should be stored according to the conditons specifed on the label.
Even repeat offenders are punished with no more than 3 years in prison or a fne of $10 cheap chloramphenicol 500 mg with amex,000 (Donaldson generic chloramphenicol 500 mg free shipping, 2010). Considering that the proft margin for falsifed drugs runs in the billions, the risk-to-proft Copyright © National Academy of Sciences. He ofered more than 40 products including Viagra, Zoloft, Lipitor, Cialis, and Xanax (Kake. His customer service call center was in the Philippines; he paid his employees through wire transfers from Costa Rica, the Philippines, and the United States. Calvelo was sentenced to 48 months in prison and, as part of his plea, agreed to pay $1. Table 4-5 shows the penalties for falsifying medi- cines in a selection of countries. Tables 4-6 and 4-7 show penalties for patent and trademark infringement, which are dealt with more severely in some countries. Stricter and more consistent penalties could do much to fght the public health crime of producing and trading fake medicines. Chapter 7 discusses this solution in more detail, describing how a global code of practice could encourage consistent strict minimum punishments for these offenses. Much as poor-quality drugs are often both falsifed and sub- standard, some potentiating factors encourage both kinds of problems. The high demand and erratic supply of drugs, weak regulatory systems, and lack of political will contribute to the trade on both falsifed and substandard drugs. Expense and Scarcity Medicines are what economists describe as a comparatively inelastic good (Arnold, 2008); changes in the unit price of the medicine have pro- portionately little effect on the demand (Siminski, 2011). Price inelastic- ity, combined with a high relative price, make medicines a major expense for patients around the world. In the United States, health expenditures on medicine rise sharply in middle life and average between $1,000 and Key Findings and Conclusions • The demand for medicines is relatively consistent, though the sup- ply is not. A more straightforward registration and application process would reduce burdens on industry and regulators. Regulators in low- and middle-income coun- tries need training, equipment, and technology, as well as guidelines for strategic decisions about what to invest in frst. Substan- dard drug production at the New England Compounding Center happened because of insufcient clarity between state and national responsibilities. Patients and providers need accurate information about the risks, communicated in way that empowers them to take reasonable precautions to protect their safety. The cost of medicine is even more of a burden in low- and middle-income countries, where it accounts for 20-60 percent of health spending, and 90 percent of the population pays for medicine out-of-pocket (Cameron et al. Some- times the supply falters because of shortages in the raw materials, as in 2004 when increased demand for artemisinin, combined with a poor Artemesia annua harvest, drove up the price and led to stock-outs (Kindermans et al. In the United States, for example, manufacturers sometimes stop producing products with low proft margins, such as sterile injectables—inexpensive products that are complicated to make (Hoffman, 2012). Manufacturers also can lose interest in a drug after its patent expires, when revenues from the product drop (Hoffman, 2012). Although the United States has a more stable drug supply than most devel- oping countries, there have been regular shortages for the past 15 years, es- pecially among injectables, cancer drugs, and antibiotics (Hoffman, 2012). Figure 4-3 shows that although private-sector outlets have a higher percentage of drugs available than public-sector ones, there is still a great deal of unmet need. A month’s course of the lowest-priced generic ulcer medication, for example, is still more than 3 days’ wages for a low-paid government worker in much of Africa, Eastern Europe, and the Middle East (Cameron et al. Reducing the costs and increasing the availability of medicines would remove some of the fnancial incentive to produce and procure falsifed and substandard medicines. If patients had a plentiful supply of reliable, affordable medicines, there would be less need to shop at unregulated gray markets. For generic manufacturers, companies that generally run on low margins, the costs of proving bioequivalence and preparing a manufacturer’s dossier for regula- tory review can be prohibitive to market entry (Lionberger, 2008). Different regulatory authorities have different, often widely divergent, requirements for establishing bioequivalence (Mastan et al. Countering the Problem of Falsified and Substandard Drugs 164 Copyright © National Academy of Sciences. The committee believes this format could be useful to regulators and gener- ics companies in low- and middle-income countries. Harmonized applications also give regulators a common format to discuss their product registration process. Like sharing inspections and other harmonization efforts, the use of the common document increases effciency and promotes a common language among regulators. Recommendation 4-3: Regulatory authorities in low- and middle- income countries should use the International Conference on Harmoni- sation Common Technical Document format for product registration to better harmonize their procedures and reduce application costs for manufacturers. To the same end, they should also conduct joint inspec- tions and use a common inspection report.
Note : Potassium dichromate can be obtained as a primary standard reagent and hence buy 250mg chloramphenicol free shipping, standard solu- tions may be prepared determinately and stored for long periods of time purchase chloramphenicol 250 mg on line. Calculations : The quantity of Mohr’s salt required for 250 ml of the solution having a normality of 0. Procedure : Transfer 20 ml of the primary standard solution (Mohr’s salt) to the titration flask and add 20 ml of 2 N sulphuric acid. Transfer drops of the titrated solution by means of a glass rod and mix with drops of the indicator, already taken in the groove-tile. The above sequential steps give fairly accurate results because the error caused by the removal of part of the solution for the spot tests is made negligibly small. By applying the relationship between N1V1 (K2Cr2O7) and N2V2 (Mohr’s salt), the normality of the former may be calculated. Procedure : (a) Preparation of Standard K2Cr2O7 Solution : Instead of using solutions having definite normal- ity, routine industrial laboratories make use of ‘emperical solution’ which is normally expressed in terms of ‘titer for the substance determined’. For this assay, let us prepare an emperical K2Cr2O7 solution (250 ml) of such a concentration that 1 ml of the same exactly correspond to 0. Add 15 ml of concen- trated hydrochloric acid, warm the contents of the flask carefully over a sand-bath until most of the dark grains of ore get dissolved completely and only a whitish silica precipitate settles at the bottom of the flask. Add 25 ml of a mixture of sulphuric acid and phosphoric acid to the solution along with 2 drops of diphenylamine indicator and titrate the solution with K2Cr2O7 solution carefully, by adding small lots at intervals with constant shaking, until a persistant blue-violet colour appears. On reduction, the resulting cerous salt obtained is colourless in appearance and, therefore, strong solutions may be considered as self-indicating. As this concentration is very dilute for observation of the respective end-point, hence the inclusion of an appropriate indicator becomes necessary. The oxidation reac- tion involved may be expressed as follows : Ce4+ + e Ce3+ It is interesting to observe that the solutions of ammoniurn ceric sulphate possess a number of advantages over permanganate and dichromate methods discussed earlier in this chapter, viz. Thus, we have : C H N + Fe2+ → Fe (C H N ) 2+ Fe (C H N ) 3+ + e 12 8 2 12 8 2 3 12 8 2 3 Orthophenanthroline Ferrous complex Ferric complex (colourless) (red) (blue) Base Orthophenanthroline (base) dissolves rapidly in aqueous solutions of ferrous salts, thereby three moles combine with one Fe2+ ion to give a complex termed as ‘ferroin’ having an, intense red colour. Now, any strong oxidizing agent converts the ferrous to a corresponding ferric complex having a slight blue colour. Cool, filter the solution through a fine-porosity sintered-glass crucible, dilute to 1 litre mark in a volumetric flask and mix thoroughly. Wash down the inner walls of the flask with 25 ml of sodium hydroxide solution, swirl to dissolve, add 100 ml of water and mix. Calculations : Therefore, the normality of ammonium ceric sulphate solution may be expressed as follows : wt. Cool the contents carefully and taking adequate precautions to avoid any atmospheric oxidation. Repeat the assay without the substance being examined (blank determination) and incorporate the correction, if any. Cognate Assays A number of pharmaceutical substances and dosage forms may be determined by the help of ceric sulphate titration methods as given in Table 6. Discuss the various theoretical aspects involved in the assay of permanganate, dichromate and ceric sulphate titration methods. The ‘permanganate methods’ essentially consist of three ways to assay pharmaceutical substances : (i) Direct titration method, (ii) Indirect titration method, and (iii) Residual titration method. Discuss direct titration method using ‘dichromate method’ in the assay of ‘Iron Ore’ with reference to the following aspects : (i) Preparation of 0. Iodimetry is a procedure based on the following reversible reaction : 2I– I + 2e 2 Hence, it can be utilized for the quantitative estimation of reducing agents like arsenites (H3AsO3) and thiosulphates (Na2S2O3) by employing a standard solution of iodine. Iodometry is an indirect procedure based on the aforesaid reversible reaction whereby the assay of oxidizing agents, for instance : ‘available chlorine’ in bleaching powder, cupric and ferric salts may be carried out by reducing them with an excess potassium iodide thereby liberating an equivalent quantity of iodine which can be estimated using a standard solution of thiosulphate. Iodimetric Assays : (a) Direct titration with iodine, (b) Residual titration method : i. Dissolve the contents of the beaker with the help of a glass rod and frequent swirling. Note : Stock solutions of sodium thiosulphate may be preserved by the addition of a few drops of sodium hydroxide solution (20% w/v) which serves as stabilizer as well as prevents decomposition. The resulting mixture is boiled gently for a further period of 30 minutes till a transluscent and thin liquid having an uniform consistency is obtained. Note : (1) The prepared solution of starch undergoes rapid deterioration, hence it is always desired to use freshly prepared solution every day, (2) It is now more or less believed that the iodine is held as an ‘absorption complex’ within the helical chain of the macromolecule βββββ-amylose i. However, another component, ααααα-amylose, is undesirable because it produces a red-colouration with io- dine which is not readily reversible, and (3) ‘Soluble Starch’ comprises principally of βββββ-amylose, with the ααααα-fraction having been removed. Always, it is a practice to prepare indicator-solutions from this product exclusively.
